Over 3,000+ companies globally trust us
From startups to global enterprise giants, Certvalue empowers to extend the breadth and depth of their customer relationships








Get Instant Quote
Certvalue is professional ISO 13485 Consultant in Afghanistan, providing ISO 13485 Certification in Afghanistan, Kabul, Jalalabad, Kandahar, Mazari Sharif, Herat, Charikar, Taleqan with the assurance of training, Audit, Documentation, Certify, Templates, Gap Analysis, Implementation, Registration, Consulting services at affordable cost to all organizations to get certified under medical device system in Afghanistan.

Professional Consulting Company
CONSULTATION & CERTIFICATION
ISO 13485 certification in Afghanistan is an industry specific standard which has been published by international organization for standardization in order to increase the process efficiency on increasing the quality of the product being manufactured. This is a standard which has been derived from the quality management system. The difference between the quality management system and quality management system for medical devices that this standard focuses more on process in manufacturing area. This standard will be applicable for manufacturers, importers, distributors and supply chain. This standard has been frequently implemented across Medical and Pharmaceutical manufacturing industries and which is one of the critically sectors compared to other industrial sector. ISO 13485 Certification in Afghanistan specifies the requirement of Management commitment involvement in order to drive the seriousness within the company in order to adopt the management system without which the results are not guaranteed and its waste of time in implementing the standard requirement. To initially start with it is always advised to conduct a gap analysis based on standard requirement, company requirement, customer requirement and legal requirement which will make you understand the gaps between the four areas. Conducting the stringent and strong gap analysis will lead to identification of hidden loopholes within the system which will impact on the quality of product being manufactured. ISO 13485 Certification in Afghanistan specifies the requirement to have a gap analysis report based on the analysis conducted with which you can plan how to start the required documentation and evidence to fulfill the standard requirement.
ISO 13485 registration services in Kandahar specify the mandatory requirement on framing the quality policy on medical manufacturing process involving all the needs and Expectations of interested parties and standard requirement which has to be approved by the top management. Post approval the policy has to be circulated internally and externally. Department objective has to be framed based on the policy is framed which is one of the next mandatory area in order to achieve the effective results. During the audit it is necessary that you the demonstrate the evidence for the objectives but in order to need a standard requirement failing to which will result in cancellation of your certification. ISO 13485 registration in Afghanistan specifies their requirement appointment of leader which has replaced the term management representative in the previous version of the standard. Quality manual is no longer a mandatory requirement from a standard but all your process and work instructions at to be documented within the standard operating procedure. Based on the Standard operating procedures you should demonstrate the evidence required for the same. The term documents and record has been clubbed which is now known as document information to avoid the confusion which used to happen during the previous version of the standard.
The Consultants of ISO 13485 certification Services in Afghanistan should be the right person having the knowledge on the Standard requirement and was the experience on the previous implementing techniques should be helpful to help you as am working hand to adopt a management system helping you to save time and cost. Standard specifies to provide the required resource in order to implement the management system. Providing training to the resource on the Standard requirement would be very essential in order to setup the successful system in place. The standard specifies to conduct frequent internal audit and management review meeting which act as a tool in measuring the improvement. The Consultants of ISO 13485 Certification Services in Afghanistan should help you to provide the competency on how to conduct the audit and how to write the audit report. It should be made sure that the consultant provides the certification after completion of training which will act as an evidence for you to demonstrate during the external audit. Bengal conducting the internal audit the leader should plan on when to conduct the internal audit by fixing the audit calendar which act as an evidence to demonstrate the system. Post finalizing the audit calendar you should finalize on the audit schedule which is nothing but who is auditing on which department. Cross department audit is always suggested in order to achieve a successful results. On completion of the internal audit all the observations should be discussed in front of everyone as this is a practice done in order to achieve the improvement. All the observations should be noted down in the audit report on which the corrective action has to be taken place. ISO 13485 audit services in Afghanistan from a third party certification body which has an accredited accreditation body will add weight age to your management system and desertification. During the audit the leader should demonstrate the evidence to the auditor on the set up management system.
What are the advantages from the standard?
- ISO 13485 Certification in Afghanistan will help you to identify all the risk involved with in the process. Identify all the risk it will help you to mitigate them in order to safeguard the business in future.
- ISO 13485 consultants in Kabul will help the company to adopt the international standards from the best practices which are being implemented across all the successful companies.
- ISO 13485 Consulting Services in Kandahar increase the confidence in both customer and employees eliminating the problems of employee retention and increasing the profit margin by hitting business from the existing customers only.
- ISO 13485 consultants in Kabul will act as an inbound marketing in attracting customers towards your product being manufactured which helps in reducing me marketing expenses and increasing the profit margin.
Now Let us know, what is the definition of the medical devices, it is one of the devices, instrument, apparatus, machine, implant or others similar article that are used for the diagnosis, prevention and treatment of the diseases or other medical conditions:
And there are different varieties of medical devices starting from Complex computer control Machines to hard tools which include:
- Simple devices like wound dressings, scalpels.
- Durable devices like wheelchair, dentist chair, hospitalized bed.
- Implantable devices like cardiac pacemakers, monitors, prosthetic joint, prosthetic limbs.
- Life supporting devices like ventilators for respiratory and lungs.
- Sophisticated, Software control devices such as MRI machines and CT scanners etc.
so for all of these devices it should have met with ISO 13485 certification in Afghanistan because of these requirements of the standard when it is applied to these devices it will be more qualitative and will be more effective when they are used.
How to get certified for ISO 13485 certification in Afghanistan?
We are one of the one stop solution providers for all of the international certification available in market. We are one of the market leaders on consultation and certification of both product and process certification on management system published by international organization for standards and other international standards which are made available in market. We are one of the award winner company having hundred percent track record of success in helping our customers to adopt the management system and helping them to achieve the intended results from the standard and the benefits of industry best practices in order to streamline the process and increase the efficiency. With us ISO 13485 certification cost in Afghanistan will remain feasible and minimal for all of our customers across the world which makes us unique compared to others. We help our customers to achieve the continual improvement by providing assistance even after the audit services.
How to get ISO 13485 Certification Consultants in Afghanistan?
If you have a requirement to get your business certified and thinking how to get ISO 13485 certification in Afghanistan, you do not have to worry for on this as you can leave this requirement to our consultants who have expertise in every industrial sectors in order to help your company to adopt the management system. You can just write to us at [email protected] or visit our official website at www.certvalue.com you know more about us and our services being delivered cross 30 different countries. You have to just register your certification requirements 2 hour help desk personal so that one of our Consulting experts will contact you at the earliest to understand about your company process and the certificate in order to help your business get certified.
Quick Enquiry Form
Our Services
- ISO 9001 Certification
- ISO 14001 Certification
- OHSAS 18001 Certification
- ISO 22000 Certification
- ISO 27001 Certification
- ISO 13485 Certification
- ISO 17025 Certification
- ISO 45001 Certification
- ISO 22301 Certification
- ISO 27701 Certification
- ISO 20000-1 Certification
- ISO 27017 Certification
- ISO 27018 Certification
- ISO 50001 Certification
- ISO 55001 Certification
- ISO 21001 Certification
- ISO 28000 Certification
- ISO 27032 Certification
- ISO 15189 Certification
- ISO 22716 Certification
- ISO 22483 Certification
- CE mark Certification
- HACCP Certification
- HALAL Certification
- SA 8000 Certification
- ROHS Certification
- BIFMA Certification
- GMP Certification

Asked Any Questions
The certificate is issued from a premium certification body and it can be verified from its -website. Every certificate comes with a unique certification number.
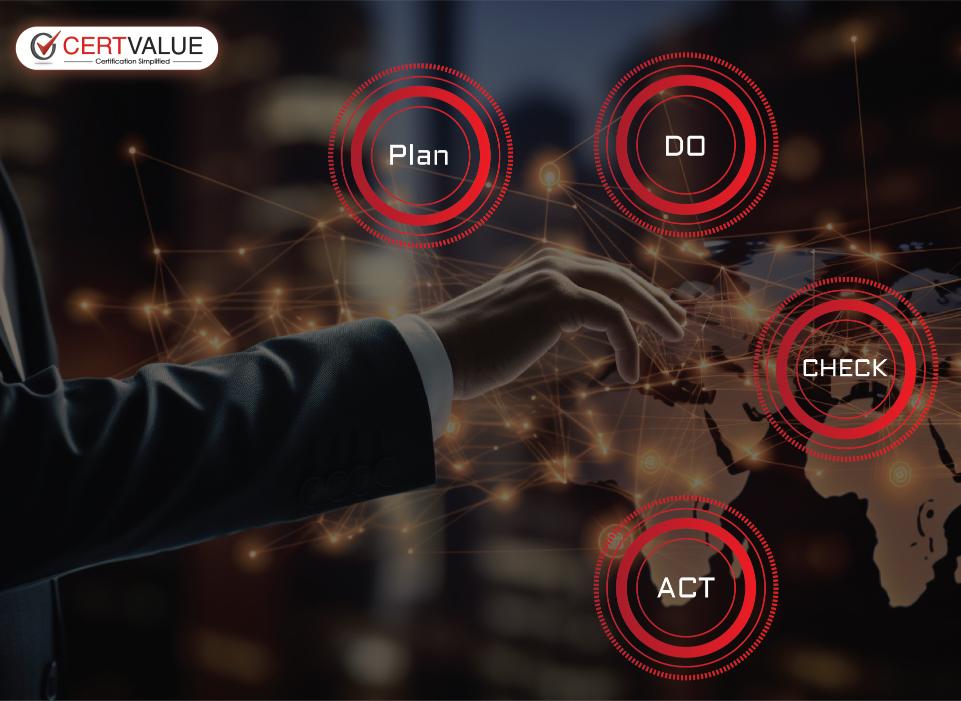
PLAN
Conduct Gap Analysis to find any Shortcomings from the standard requirements.
DO
Policies, procedures, Work Instructions, Evidences, Records, Training
CHECK
Conduct frequent internal audit and management review meeting.
ACT
Apply corrective actions on the identified root cause or shortcomings
Process for ISO Certification in Bangalore
Certvalue make the ISO certification process in a simple way for every organization in bangalore to get their certification sitting at their place in lesser time and at an affordable cost.
Get Consultation
Conduct Gap Analysis to find any Shortcomings from the standard requirements.
Submit Your Documents
Policies, procedures, Work Instructions, Evidences, Records, Training
Get Audited
Undergo a thorough audit by Certvalue’s auditors to assess compliance.
Get ISO Certificate
Upon successful audit, receive your ISO certification from Certvalue.

WHY CERTVALUE?
CERTVALUE – CREATING VALUE FOR YOUR CERTIFICATION

Client/Compliance
Bottom-line of any business organization is profit and Customers are the only source of Profit. Certvalue will help balancing both customer and compliance requirement at the same time with the help of ISO certification

Enhancement of Performance
ISO certification is a tool to streamline and enhance the process performed internal to the organization. Certvalue indulges in inculcating best industry practices.

Recognition and Brand Value
It is always about the Brand value of your organization in the market and ISO certification from Certvalue can make your organization to be an excel and stand out in the market globally

Tender Eligibility
ISO certification is a basic requirement to bid or participate in any tenders floated by government or private sector. And ISO certification from Certvalue is an assurance win over the tenders.
Extract all the benefits of our quality consultation & implementation
Partner Us to achieve the greatest Accomplishments !!
Our Client Reviews















